- Home
- Steam Resources
- Steam Theory
- Vacuum Steam Heating Systems
Steam Control
Vacuum Steam Heating Systems
What is a Vacuum Steam Heating System?
When plants require a temperature such as 60°C [140°F] or 90°C [194°F] for an indirect heating application, they often choose to heat with circulated hot water. However, heating in these temperature ranges is also possible with steam. As mentioned in Vacuum Steam Basics, if the pressure of saturated steam is reduced below atmospheric pressure, its temperature can be lowered to, for example, 60°C or 90°C.
Vacuum steam heating systems are systems that use saturated steam below atmospheric pressure as a heating medium. Compared with other heat technologies, vacuum steam heating systems have the following advantages.
- Even heating
- Quicker heating compared to hot water
- Potentially smaller temperature difference between the product and the heating medium, reducing burns and product damage
- Small equipment footprint
Difference Between Vacuum Steam and Hot Water Heating
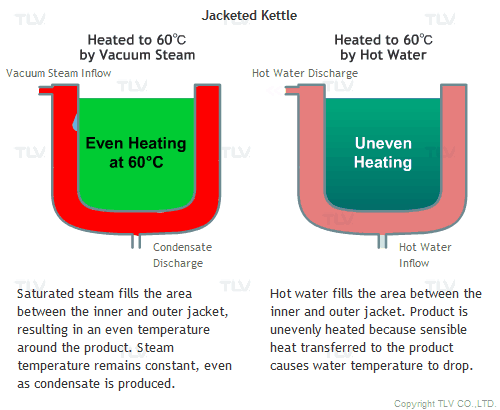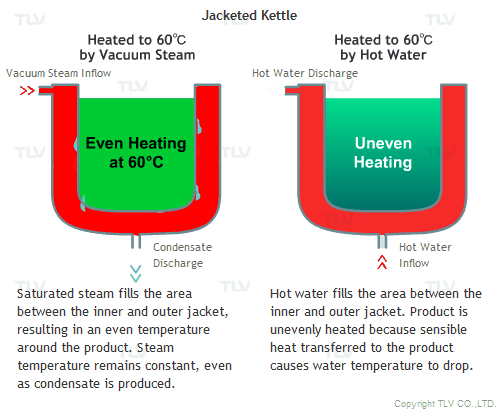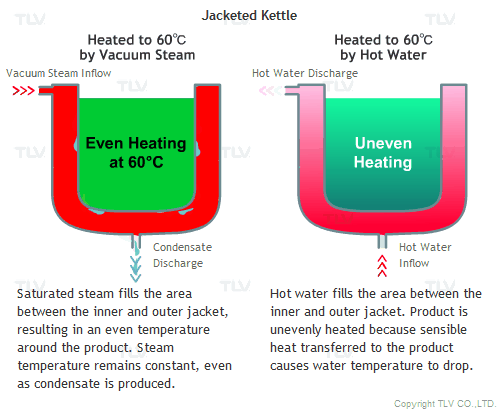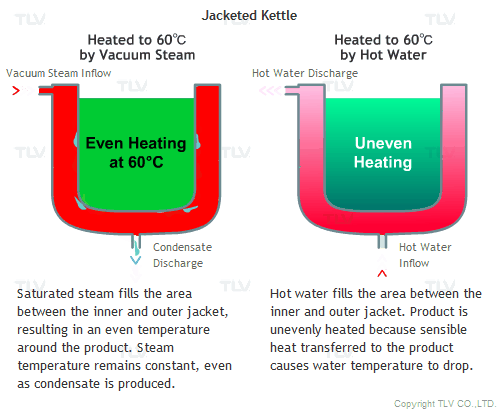
All the advantages mentioned above assume that vacuum steam is saturated. Since it is saturated steam, the temperature of the space filled with steam is uniform, and heat is transferred quickly by latent heat from steam to the product.
Hot water heating, on the other hand, transfers heat to the product by sensible heat, and by doing so, the temperature of the hot water itself will decrease. This can be observed by measuring the temperature at the hot water inlet and outlet. To minimize this temperature difference and to supply the required amount of heat quickly, an enormous amount of hot water needs to be supplied.
Overall heat transfer coefficient
This section is an excerpt of the full article: Overall Heat Transfer Coefficient. See the article for a more thorough explanation.
The overall heat transfer coefficient, or U-value, refers to how well heat is conducted through over a series of resistant mediums. Its units are the W/(m²°C) [Btu/(hr-ft²°F)].
The overall heat transfer coefficient is influenced by the thickness and thermal conductivity of the mediums through which heat is transferred. The larger the coefficient, the easier heat is transferred from its source to the product being heated. In a heat exchanger, the relationship between the overall heat transfer coefficient (U) and the heat transfer rate (Q) can be demonstrated by the following equation:
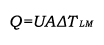
where
- Q = heat transfer rate, W=J/s [btu/hr]
- A = heat transfer surface area, m2 [ft2]
- U = overall heat transfer coefficient, W/(m2°C) [Btu/(hr-ft2°F)]
- ΔTLM = logarithmic mean temperature difference, °C [°F]
From this equation it can be seen that the U value is directly proportional to Q, the heat transfer rate. Assuming the heat transfer surface and temperature difference remain unchanged, the greater the U value, the greater the heat transfer rate. In other words, this means that for a certain heat exchanger and product, a higher U value could lead to shorter batch times and increased production/revenue.
For more information on the overall heat transfer coefficient and how to calculate U values, refer to the full article here: Overall Heat Transfer Coefficient
The next article in our series on steam control will move away from heating, and focus on cooling applications for vacuum steam.